Implementing Change Control Management in ISO 13485
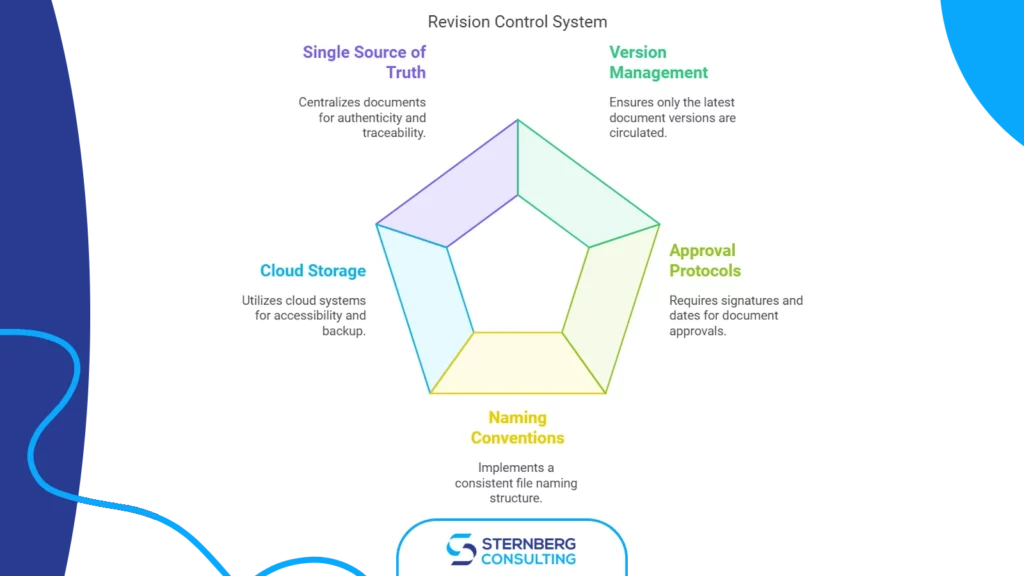
Change control is one of the most critical processes in an ISO 13485 quality management system for medical devices. Learn how to implement a robust change control procedure that meets regulatory requirements, manages risk, and keeps your documentation aligned with actual product and process changes.

