Implementing Change Control Management in ISO 13485
Change control is one of the most critical processes in an ISO 13485 quality management system for medical devices. Learn how to implement a robust change control procedure that meets regulatory requirements, manages risk, and keeps your documentation aligned with actual product and process changes.
Managing Change Control in ISO 13485

ISO 13485 requires organizations to systematically control changes to processes, products, and documentation. This guide covers the core requirements for change control management, how to build a practical change request workflow, and what auditors will look for during certification.
Feedback Instead of Complaints? The Key Differences

Complaints and feedback are not the same thing — and treating them identically can cost you valuable customer insight. This article explains the key differences, why the distinction matters for your quality management system, and how to structure a process that captures both effectively.
ISO 13485 Clause 4.1: General Quality Management System Requirements

Review ISO 13485 – Clause 4.1 to get familiar with General QMS Requirements for companies in the medical device industry. We’ll cover documentation needs, processes, risk management, and much more.
Check Sheet – Data Collection Tool

A check sheet is one of the seven basic quality tools — a simple, structured form for collecting and recording data in real time. Learn when to use check sheets, how to design them effectively, and how they support root cause analysis and process improvement within a quality management system.
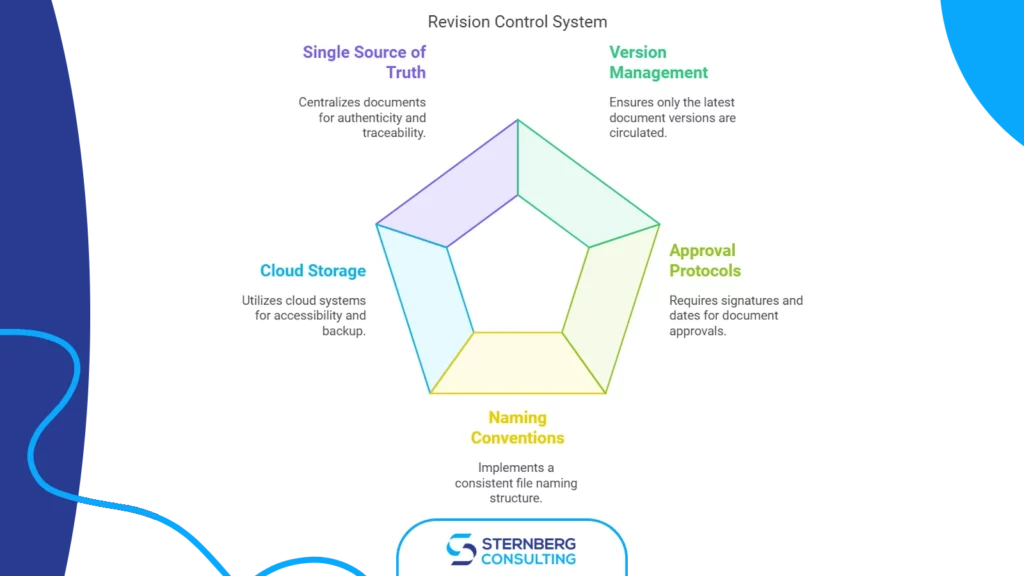
Efficient ISO 9001 Document Control Solutions

Document control is a core ISO 9001 requirement and one of the areas most frequently cited in audits. This guide covers practical approaches to managing controlled documents, version histories, and approval workflows — whether you use a simple shared folder or a dedicated document management system.
Box and Whisker Plot – Displaying Variation in Data

A box and whisker plot is a powerful visual tool for displaying the distribution and variability of a data set at a glance. Understand how to read and construct box plots, what the quartiles and whiskers represent, and how this statistical tool supports data-driven quality management decisions.
Free ISO 9001 Work Instruction Template for Effective Documentation

Work instructions are the most detailed level of your QMS documentation — describing exactly how specific tasks should be performed. Download a free, ready-to-use ISO 9001 work instruction template and learn how to write instructions that are clear, practical, and consistently followed on the shop floor.
ISO 13485 Design Control: Best Practices

Design control is one of the most complex and audit-critical requirements in ISO 13485 for medical device manufacturers. This guide explains the design and development lifecycle requirements — from planning and inputs through verification, validation, and transfer — with practical best practices for documentation and traceability.
Attribute and Variable Data – Types of Quality Data

Understanding the difference between attribute data and variable data is fundamental to selecting the right statistical methods in quality management. This guide explains both data types, when each applies, and how they influence which control charts, sampling plans, and analytical tools you should use.

